Rotavirus in infants
What is RotaVirus? Symptoms, Causes,risk factors, types of vaccines available
What Is Rotavirus?
Rotavirus is a virus that causes diarrhea and other intestinal symptoms. It’s very contagious and is the most common cause of diarrhea in infants and young children worldwide. If you look at a rotavirus through a microscope, it has a round shape. The Latin word for wheel is “rota,” which explains how the virus got its name.
Rotavirus causes inflammation in the stomach and intestines. It can cause severe diarrhea, vomiting, fever, belly pain, and dehydration in infants, young children, and some adults.
Medications can help with the symptoms, but there’s no cure for rotavirus. Even children who have been vaccinated against it may get it more than once.
Rotavirus Symptoms
If your child has come into contact with rotavirus, symptoms won't show up for about 2 days. Then, they’ll have:
- Fever, vomiting, and stomach pain. Rotavirus usually starts with these symptoms, which then fade away.
- Diarrhea begins after the first three symptoms have stopped. As the virus works its way through your child's system, the diarrhea can hang on for 5 to 7 days.
Call your doctor if your child has:
- Lethargy
- Frequent vomiting
- Less desire to drink fluids
- Stools that are black or contain blood or pus
- Any high temperature in a baby younger than 6 months
- A high temperature for more than 24 hours in a child older than 6 months
With all the vomiting and diarrhea, your child may not feel like eating or drinking. This can make them dehydrated, which might even become life-threatening. Older adults, especially those with other illnesses or conditions, could also get dehydrated.
Call your doctor if you notice any of these symptoms of dehydration:
- Anxiousness
- Crying with no tears
- Little urination or dry diapers
- Dizziness
- Dry and throat
- Severe sleepiness
- Pale skin
- Sunken eyes
Adults often have similar symptoms, but they tend to be less severe.
Rotavirus Causes and Risk Factors
Anyone can get rotavirus, but it most commonly affects:
- Infants
- Young children
- Close relatives
- Those who work with children, such as nannies or child care workers
If your child has rotavirus, it's in their poop before symptoms start and up to 10 days after they taper off. During that time, when your child wipes after using the toilet, rotavirus can spread to their hands. If they don't wash their hands, they might contaminate anything they touch, including:
- Crayons and markers
- Food
- Surfaces such as sinks and kitchen counters
- Toys, including shared electronics such as iPads and remote controls
- Utensils
- Water
If you touch your child's unwashed hands or any object they’ve contaminated and then touch your mouth, you can be infected.
Disinfecting is key. Rotavirus can live on surfaces and objects for weeks.
Rotavirus vaccine
It is a vaccine used to protect against rotavirus infections, which are the leading cause of severe diarrhea among young children. The vaccines prevent 15–34% of severe diarrhea in the developing world and 37–96% of severe diarrhea in the developed world. The vaccines decrease the risk of death among young children due to diarrhea. Immunizing babies decreases rates of disease among older people and those who have not been immunized.
The vaccine first became available in the United States in 2006. It is on the World Health Organization's List of Essential Medicines. As of 2013, there are two types of vaccine available globally, Rotarix and RotaTeq. Others are used in some countries.
Rotarix
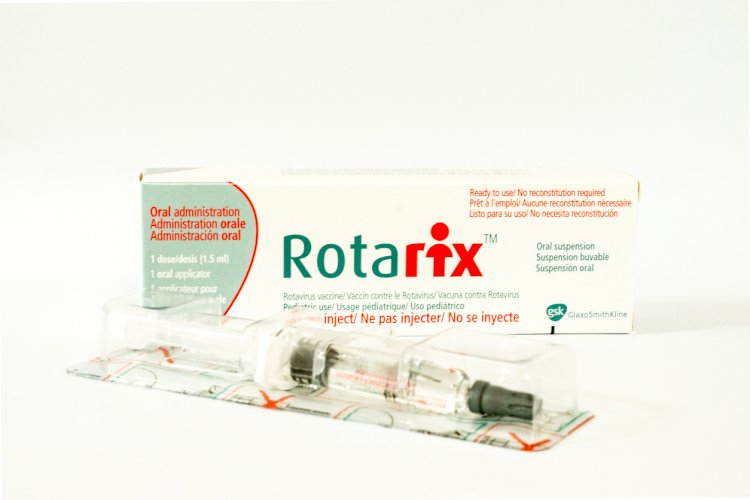
Rotarix is a monovalent, human, live attenuated rotavirus vaccine containing one rotavirus strain of G1P[8] specificity. ROTARIX is indicated for the prevention of rotavirus gastroenteritis caused by G1 and non-G1 types (G3, G4, and G9) when administered as a 2-dose series in infants and children. It was approved by the U.S. FDA in April 2008.
RotaTeq

RotaTeq is a live, oral pentavalent vaccine that contains five rotavirus strains produced by reassortment. The rotavirus A parent strains of the reassortants were isolated from human and bovine hosts. Four reassortant rotaviruses express one of the outer capsid, VP7, proteins (serotypes G1, G2, G3, or G4) from the human rotavirus parent strain and the attachment protein VP4 (type P7) from the bovine rotavirus parent strain. The fifth reassortant virus expresses the attachment protein VP4, (type P1A), from the human rotavirus parent strain and the outer capsid protein VP7 (serotype G6) from the bovine rotavirus parent strain. In February 2006, the U.S. Food and Drug Administration approved RotaTeq for use in the United States. In August 2006, Health Canada approved RotaTeq for use in Canada. Merck worked with a range of partners including governmental and non-governmental organisations to develop and implement mechanisms for providing access to this vaccine in the developing world, an effort which was slated to come to an end in 2020
What's Your Reaction?














































































